Oral Vaccine for HIV Immunization
Application
Live bacteria-based vaccine that is delivered by oral route and stimulates the body's immune system to produce mucosal antibodies against HIV.
Key Benefits
- Provides better vaccine scheme via mucosal immune response as a primary vaccine compared to other primary vaccines that are administered parenterally.
- Generates both antibody and T-cell responses.
- First-in-class treatment that could be combined with other approaches as well.
- Does not need refrigeration and is inexpensive to produce.
- Good safety profile already exists concerning human oral ingestion of L. lactis.
Market Summary
The majority of HIV infections are contracted via mucosal routes; therefore, a vaccine that elicits strong HIV-specific immunity at the mucosal tissue would restrict virus replication very early at the site of initial virus exposure and optimally enhance protection from infection. Generally, immunizations through mucosal routes have elicited far better responses in mucosal tissues such as the intestines, than the systemic route. Most HIV vaccines under development are delivered intramuscularly, which is relatively poor for generating strong, long-lived mucosal immune responses. Only one vaccination in clinical trials utilizes a mucosal prime and it is administered intranasally. None of the current technologies are administered orally.
Technical Summary
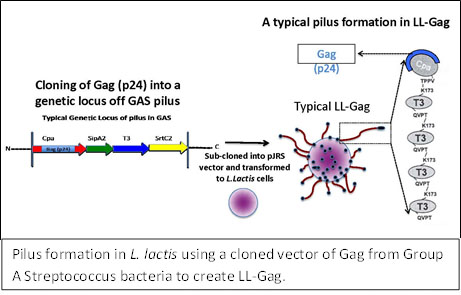
While the preferred route of administration of a mucosal immunity vaccine would be by mouth, an orally delivered vaccine must be able to survive the high acidity in the stomach in order to elicit an immune response in the intestinal tract. Emory researchers are developing a vaccine that enhances induction of HIV-specific humoral and cellular immunity using a bacterium that can survive in the acidic environment of the stomach. This mucosal vaccine consists of modified Lactococcus lactis, a type of bacteria commonly used to produce yogurt. Although L. lactis bacteria do not normally express long arm-like structures called pili, the bacteria for this vaccine have been engineered to contain pili (described in tech ID 08099) and express HIV proteins at the ends of these pili. Because the bacteria naturally survive the acid environment of the stomach and can live for days in the intestinal tract, the vaccine can be taken orally. Oral immunization with this therapy in mice elicits strong IgG and IgA responses in mucosal secretions and serum. In addition, combining this oral immunization with an intramuscular boost from a recombinant viral vector-based vaccine produces systemic and intestinal Gag-specific CD8 T cell responses. In initial studies in rhesus macaques, immunizations elicit a strong Gag-specific CD4 T cell responses in the rectum. Unlike most existing vaccines, the Lactococcus-based vaccine does not require cold storage and is inexpensive to produce.
Developmental Stage
L. lactis expressing Gag as an antigen has been constructed and tested in mice and rhesus macaques.
Patent Information
| App Type |
Country |
Serial No. |
Patent No. |
File Date |
Issued Date |
Patent Status |
| Nationalized PCT - United States |
United States |
14/422,490 |
9,861,693 |
2/19/2015 |
1/9/2018 |
Issued |
| Divisional |
United States |
15/848,706 |
10,729,762 |
12/20/2017 |
8/4/2020 |
Issued |
|
|

