Microfluidic Device for Increased Viral Transduction Efficiency
Application
Microfluidic chip device for facilitating high throughput viral transduction for gene therapy applications.
Key Benefits
- Improves surface-to-volume ratio, allowing more efficient contact between lentiviruses and cells
- Increases efficiency while maintaining cell viability of traditional methods.
- Enables gene transfer for non-adherent cells.
- Compatible with syringe pump.
Market Summary
Lentiviral vector gene therapy is one of the predominant methods used for gene transfer during gene therapy applications. Although lentiviral vectors are very effective mechanism for gene transduction, one of the key challenges to their use is that they require large quantities or a high concentration of virus to ensure transfer into most cells. Given that generating the lentiviral vectors used for gene therapy is expensive, this potentially makes the application of lentiviruses for gene therapy cost prohibitive regardless of their effectiveness. Current techniques for performing viral transfection for non-adherent cells result in low transfection rates and poor cost/benefit ratios.
Technical Summary
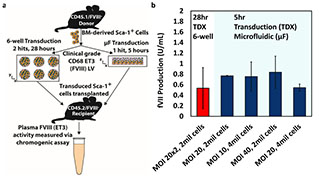
Lentiviral vector gene therapy is a method by which genes can be inserted, modified, or deleted in organisms using modified lentiviruses. Dr. Lam and colleagues have designed a microfluidic device to improve the efficiency of lentiviral gene transfer, particularly for use when modifying common non-adherent target cell types such as hematopoietic stem cells. The microfluidic device yields a larger surface area to volume ratio and immobilizes these cells in a channel lined with retronectin. The device can be easily modified to accommodate cells of different sizes or different channel geometries. This device can improve the efficiency of lentiviral gene transfer and may be used in creating induced pluripotent stem cells (iPSCs), RNAi therapy delivery, and gene therapy.
Developmental Stage
- Testing of T-cell transduction conducted in clinically relevant cell type.
- Prototype under development.
Publication: Tran, R., et al. Biomicrofluidics. 2014 Jul 30;8(4):046501.
Read our featured innovation.
Patent Information
| App Type |
Country |
Serial No. |
Patent No. |
File Date |
Issued Date |
Patent Status |
| Continuation |
United States |
16/696,456 |
|
11/26/2019 |
|
Pending |
|
|