Layer-by-layer Assembled Nanoparticles for Oral Drug Delivery
Application
Layered, casein coated magnetic nanoparticles for oral delivery of drugs protected from acid degradation.
Key Benefits
- Enables oral delivery of hydrophobic drugs that are sensitive to acid degradation in stomach condition.
- Replaces injections and infusions, reducing side effects and improving patient’s quality of life.
- Allows magnetic resonance imaging (MRI) of drug distribution and potentially magnetic guided delivery.
Market Summary
Oral delivery is the preferred route of drug administration, yet many drugs must be administered via injection or IV infusion. Oral delivery increases patient compliance to adhere to the regime of prescription drugs and improves the quality of patient life by replacing repetitive infusions or injections of drugs. Currently, doxorubicin must be administered via IV infusion in all preparations, requiring patients to visit a treatment center every few weeks. Oral delivery would improve the patient’s quality of life during chemotherapy as well as reduce the number of side effects due to infusions.
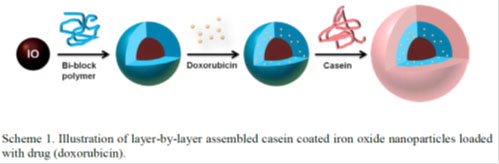
Technical Summary
Emory inventors have developed a composition enabling oral delivery of drugs susceptible to acid degradation. The composition uses an iron oxide nanoparticle core encapsulated with a bi-block polymer loaded with drugs, such as doxorubicin. This is wrapped in casein, which significantly reduces degradation when exposed to physiological levels of pepsin and pH in the gastric environment. When exposed to the physiological levels of trypsin and pH of intestines, more of the casein coat is lost. This composition reduces the pre-mature drug release in the acidic stomach and allows the enzymatic-responsive release in the small intestine, achieving small intestine targeted drug delivery. This nanoparticle construct also serves as a magnetic resonance imaging (MRI) contrast agent, providing the potential for MRI monitored and/or magnetic directed drug delivery.
Developmental Stage
Oral administration during in vivo imaging of mice show increased MRI and near infrared spectroscopy signal in the small intestine compared to non-coated nanoparticles.
Patent Information
| App Type |
Country |
Serial No. |
Patent No. |
File Date |
Issued Date |
Patent Status |
| Utility (parent) |
United States |
14/882,482 |
9,737,492 |
10/14/2015 |
8/22/2017 |
Issued |
| Divisional |
United States |
15/672,666 |
10,537,531 |
8/9/2017 |
1/21/2020 |
Issued |
|
|